The following procedures are contraindicated for patients with a deep brain stimulation system. This system is contraindicated for patients who meet the following criteria: Unilateral or bilateral stimulation of the ventral intermediate nucleus (VIM) of the thalamus for the suppression of disabling upper extremity tremor in adult essential tremor patients whose tremor is not adequately controlled by medications and where the tremor constitutes a significant functional disability.Bilateral stimulation of the subthalamic nucleus (STN) or the internal globus pallidus (GPi) as an adjunctive therapy to reduce some of the symptoms of advanced levodopa-responsive Parkinson’s disease that are not adequately controlled by medications.The Abbott Medical deep brain stimulation system is indicated for the following conditions: The system is intended to be used with leads and associated extensions that are compatible with the system. This neurostimulation system is designed to deliver low-intensity electrical impulses to nerve structures. 2021.ĭeep Brain Stimulation (DBS) Therapy Intended Use NeuroSphere™ Virtual Clinic Neuromodulation Memo. 
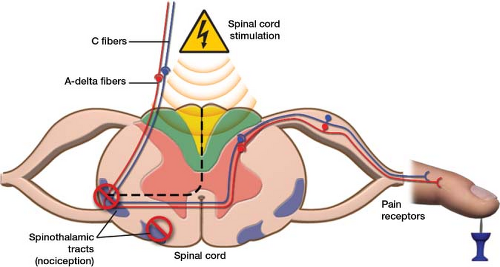
Lead and extension kits for deep brain stimulation systems. Proclaim™ DRG Neurostimulation System Clinician’s Manual. Dorsal root ganglion stimulation yielded higher treatment success rate for complex regional pain syndrome and causalgia at 3 and 12 months: a randomized comparative trial. Eterna Lowest Recharge Burden Comparison Memo (MAT-2210739). Eterna IPG Elect Design Verification Report: Current Draw (90860050). Eterna IPG Battery Recharge Characterization Report (90903492) 2022. Rat fMRI brain responses to noxious stimulation during tonic, burst, and burst-microdosing spinal cord stimulation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
